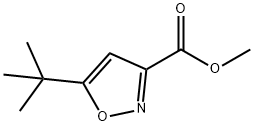
METHYL 5-TERT-BUTYLISOXAZOLE-3-CARBOXYLATE
Methyl 5-tert-butylisoxazole-3-carboxylate
CAS: 517870-22-3
Molecular Formula: C9H13NO3
METHYL 5-TERT-BUTYLISOXAZOLE-3-CARBOXYLATE - Names and Identifiers
METHYL 5-TERT-BUTYLISOXAZOLE-3-CARBOXYLATE - Physico-chemical Properties
| Molecular Formula | C9H13NO3 |
| Molar Mass | 183.2 |
| Density | 1.077±0.06 g/cm3(Predicted) |
| Boling Point | 262.4±28.0 °C(Predicted) |
| pKa | -4.93±0.50(Predicted) |
| Storage Condition | Sealed in dry,Room Temperature |
| Sensitive | IRRITANT |
| MDL | MFCD03778975 |
METHYL 5-TERT-BUTYLISOXAZOLE-3-CARBOXYLATE - Introduction
Methyl is an organic compound with the chemical formula C10H15NO3.
Nature:
-Appearance: White granular or crystalline solid.
-Melting point: about 65-68 degrees Celsius.
-Boiling point: about 295 degrees Celsius.
-Density: about 1.07 g/ml.
-Solubility: Soluble in organic solvents such as ethanol and dichloromethane.
Use:
Methyl has many uses in the chemical industry, including but not limited:
-Used as an intermediate in organic synthesis and as a raw material for the synthesis of various natural products and drugs.
-In drug research and development, it can be used as a building block of drug molecules.
-can also be used as dyes, fragrances and protective agents in the field of compound synthesis.
Preparation Method:
The preparation method of Methyl can be carried out by the following steps:
1. p-nitroaniline and ethyl isobutyl ketone react with carbon cyanide under the catalysis of alkali to generate isoxazoline compounds.
2. The resulting isoxazoline compounds are reacted with formic anhydride to obtain Methyl.
Safety Information:
- Methyl is combustible and can cause combustion when exposed to an open flame or high temperature.
-use to maintain good ventilation, avoid contact with skin, eyes and respiratory tract.
-Avoid contact with oxidants and strong acids to prevent dangerous reactions.
-Storage should be stored in a dry, cool, ventilated place, away from fire and heat sources.
Before using Methyl, it is recommended to consult the relevant safety data sheets and operating instructions in detail to ensure proper use and handling.
Nature:
-Appearance: White granular or crystalline solid.
-Melting point: about 65-68 degrees Celsius.
-Boiling point: about 295 degrees Celsius.
-Density: about 1.07 g/ml.
-Solubility: Soluble in organic solvents such as ethanol and dichloromethane.
Use:
Methyl has many uses in the chemical industry, including but not limited:
-Used as an intermediate in organic synthesis and as a raw material for the synthesis of various natural products and drugs.
-In drug research and development, it can be used as a building block of drug molecules.
-can also be used as dyes, fragrances and protective agents in the field of compound synthesis.
Preparation Method:
The preparation method of Methyl can be carried out by the following steps:
1. p-nitroaniline and ethyl isobutyl ketone react with carbon cyanide under the catalysis of alkali to generate isoxazoline compounds.
2. The resulting isoxazoline compounds are reacted with formic anhydride to obtain Methyl.
Safety Information:
- Methyl is combustible and can cause combustion when exposed to an open flame or high temperature.
-use to maintain good ventilation, avoid contact with skin, eyes and respiratory tract.
-Avoid contact with oxidants and strong acids to prevent dangerous reactions.
-Storage should be stored in a dry, cool, ventilated place, away from fire and heat sources.
Before using Methyl, it is recommended to consult the relevant safety data sheets and operating instructions in detail to ensure proper use and handling.
Last Update:2024-04-09 15:18:08
Supplier List
Spot supply
Product Name: Methyl 5-(tert-butyl)isoxazole-3-carboxylate Visit Supplier Webpage Request for quotationCAS: 517870-22-3
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 5-TERT-BUTYL-ISOXAZOLE-3-CARBOXYLIC ACID METHYL ESTER Request for quotation
CAS: 517870-22-3
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 517870-22-3
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Spot supply
Product Name: Methyl 5-(tert-butyl)isoxazole-3-carboxylate Visit Supplier Webpage Request for quotationCAS: 517870-22-3
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 5-TERT-BUTYL-ISOXAZOLE-3-CARBOXYLIC ACID METHYL ESTER Request for quotation
CAS: 517870-22-3
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 517870-22-3
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
View History

